Introduction
In vitro cell culture platforms are essential for modern research, clinical studies, and drug development. For more than a century, the petri dish has been the cornerstone of in vitro cell culture. The inventor of this technology, Julius Richard Petri, intended its use for the culture of microorganisms. However, time has proven it useful for a large variety of biological and biomedical applications. For over a century, these devices have been produced in a plethora of materials and formats and have been a groundbreaking breakthrough in biological research.
Downsize of culture platforms such as the petri dish
However, the unceasing scientific progress has shown that petri dishes do not fully reproduce the reality of in vivo organisms. In other words, this technology fails to recreate the physiological environment of cells in vivo.
When culturing cells on a plastic or glass plate, we overlook many stimuli from the cells’ in vivo microenvironment. These stimuli, however, significantly influence how cells behave and express themselves, mimicking their functions in the human body. The Spanish philosopher Ortega y Gasset once said “I am myself and my circumstances”. This is equally true for cell biology, “the cells are themselves and their microenvironment”. Failing to recreate it correctly, will be misrepresenting the behavior of cells in vivo in its entirety.
This train of thought has inspired engineers and biologists worldwide to develop new cell culture platforms addressing this unfulfilled need. Among these innovations, the most promising candidates are microfluidic devices, which aim to faithfully recreate the cells’ microenvironment.
Organ-on-chip technology
Microfluidics is the science that studies fluidic manipulation in the microscale by confining the fluidic flow in channels of dimensions in that scale (10-6-10-3m). These platforms applied to cell culture are known as “organ-on-chip” and allow the recreation of the mechanical, electrical, and chemical stimuli that cells undergo in vivo.
Beonchip (Biomimetic Environment On Chip) is devoted to the design, development and commercialization of these platforms. Our multidisciplinary team which includes, biologists, engineers, and physicists, works daily to develop new cell culture platforms and models, that represent biological processes fully and show more reliable results than classical in vitro models. Our ultimate goal is to reduce animal testing in drug development and toxicity testing by creating advanced in vitro platforms. Additionally, these platforms aim to identify new clinical biomarkers and therapies while accelerating the vaccine development process. Furthermore, the reduction of animal research in favor of in vitro studies will save time and cost for researchers and pharmaceutical companies in their R&D.
How does organ-on-chip technology recreate the physiological environment of the human body?
Firstly, using microchannels and compartments in the fluidic path allows the organized deposition of materials and specific proteins simulating an extracellular matrix. This allows the cells to adhere and interact between them as they would in vivo. Then, the tissue under study is connected to a flow of media that resembles the blood flow traversing the capillary that irrigates a biological tissue. This flow causes mechanical stress on the tissue (shear stress) and this force has a direct impact on the cell expression and morphology. Moreover, the flow indirectly influences cells by creating oxygen and nutrient gradients. As a result, these gradients can trigger phenomena like cellular migration or differentiation.
One of the greatest advantages of organ-on-chip technology is the ability to add various components to the media. For example, drugs, immune system cells, viruses, bacteria, and other microorganisms that help recreate the microbiome can be perfused through the chip. Thus, by combining different cell types in an in vivo-like architecture and applying all the physical and chemical stimuli that a tissue undergoes, we can recreate a portion of an organ or a tissue building a functional unit.
Body-on-Chip and personalized medicine
These functional units can be interconnected to simulate the crosstalk of different organs in the body, therefore, allowing the study of complex biochemical and physiological responses of this interconnected system. This concept is known as body-on-chip. It is worth mentioning an example of this technology developed by the researchers of the Wyss Institute in the USA. They connected a gut-on-chip with a liver and kidney-on-chip to evaluate the effect of orally administrated nicotine. After absorption in the intestinal wall, the substance travels through the simulated circulatory systems and is delivered to the liver. There the nicotine is metabolized and finally, it arrives in the kidney where it is excreted [1].
In the long term, this technology will be used in personalized medicine, extracting cells from a patient, and culturing them in a chip to replicate the patient’s disease in vitro. This replica of the patient will allow physicians and biologists to test different drugs and treatments to observe which of the treatments suits each patient best.
Skin-on-a-chip: A practical example
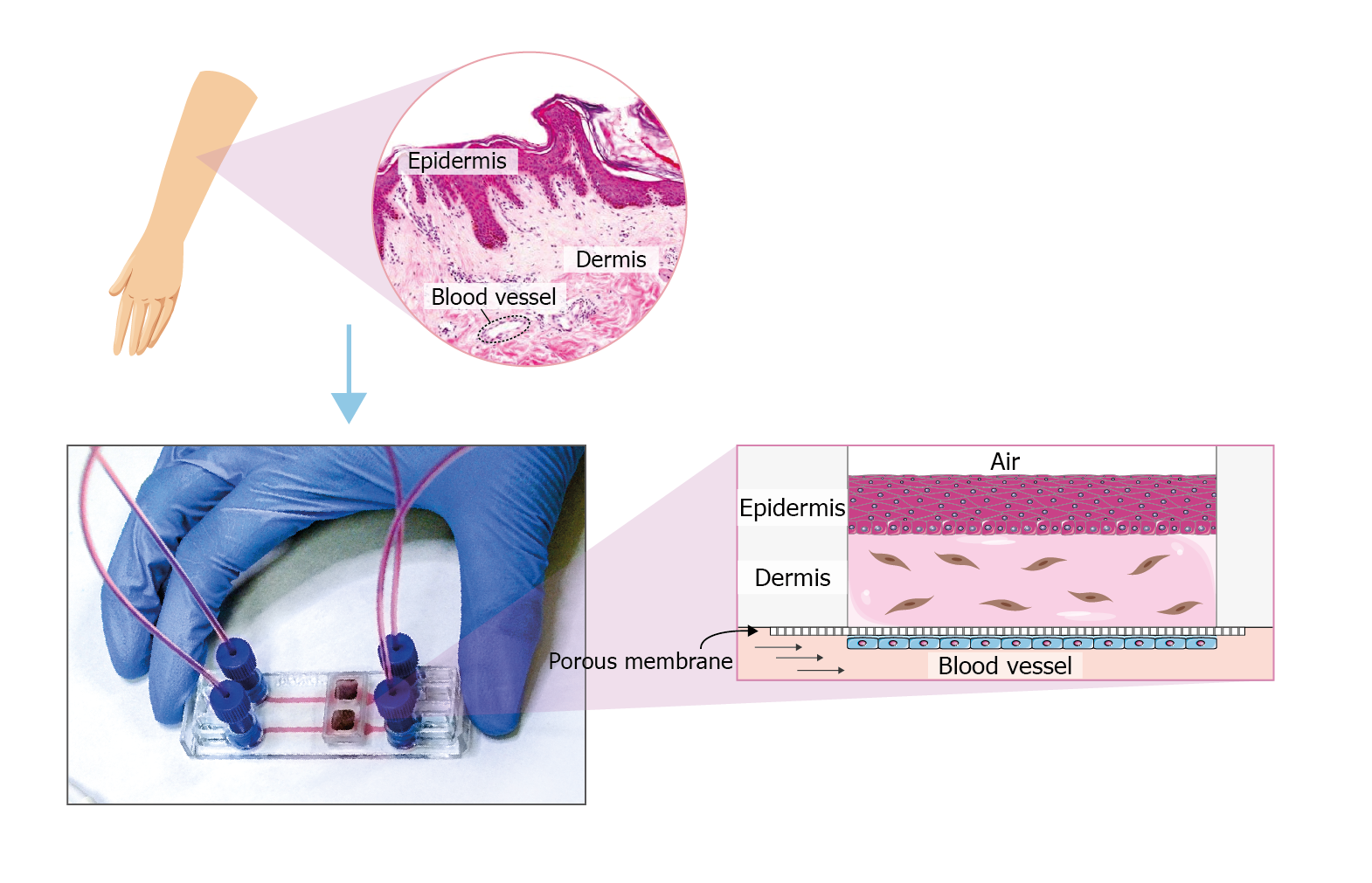
The figure shows an example of how to recreate a skin-on-chip model in the microfluidic device Be-Transflow. This device allows the study of complex culture configurations by joining a culture well with a microfluidic channel via a porous membrane. In this device, it is possible to recreate the blood vessel that irrigates the skin tissue, covering the membrane that separates the channel and well with endothelial cells. The dermis which is mainly composed of collagen and fibroblasts is simulated using a 3D culture of fibroblasts embedded in a hydrogel. After the polymerization of the hydrogel, we can culture a layer of keratinocytes in an air-liquid interface. These culture conditions will favour the growth of the different strata of the epidermis.
Future of Beonchip and the organ-on-chip technology
Our goal at Beonchip is to grow with the organ-on-chip technology and reach all of the targets mentioned in this post, helping closely our customers to adopt the new generation of cell culture platforms with the aid of our in-house R & D team. Together we will continue working to develop these innovative platforms paving the way for a more efficient and ethical biomedical research landscape.
Authors
This article was written by Sandra Gonzalez Lana and Luis Serrano.
Bibliography
- Herland, A. et al. Quantitative prediction of human pharmacokinetic responses to drugs via fluidically coupled vascularized organ chips. Biomed. Eng. 4, 421–436 (2020).
- Altas de histología. Facultad de Medicina. Universidad de Zaragoza.

