Technology
Dive into the next gen in vitro platform with our devices designed for maximum ease of use and versatility.
Materials
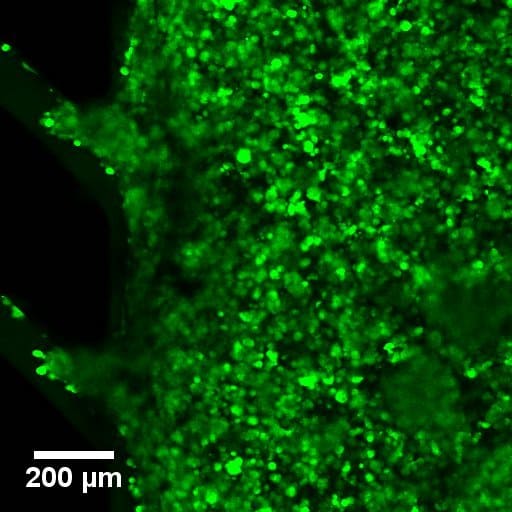
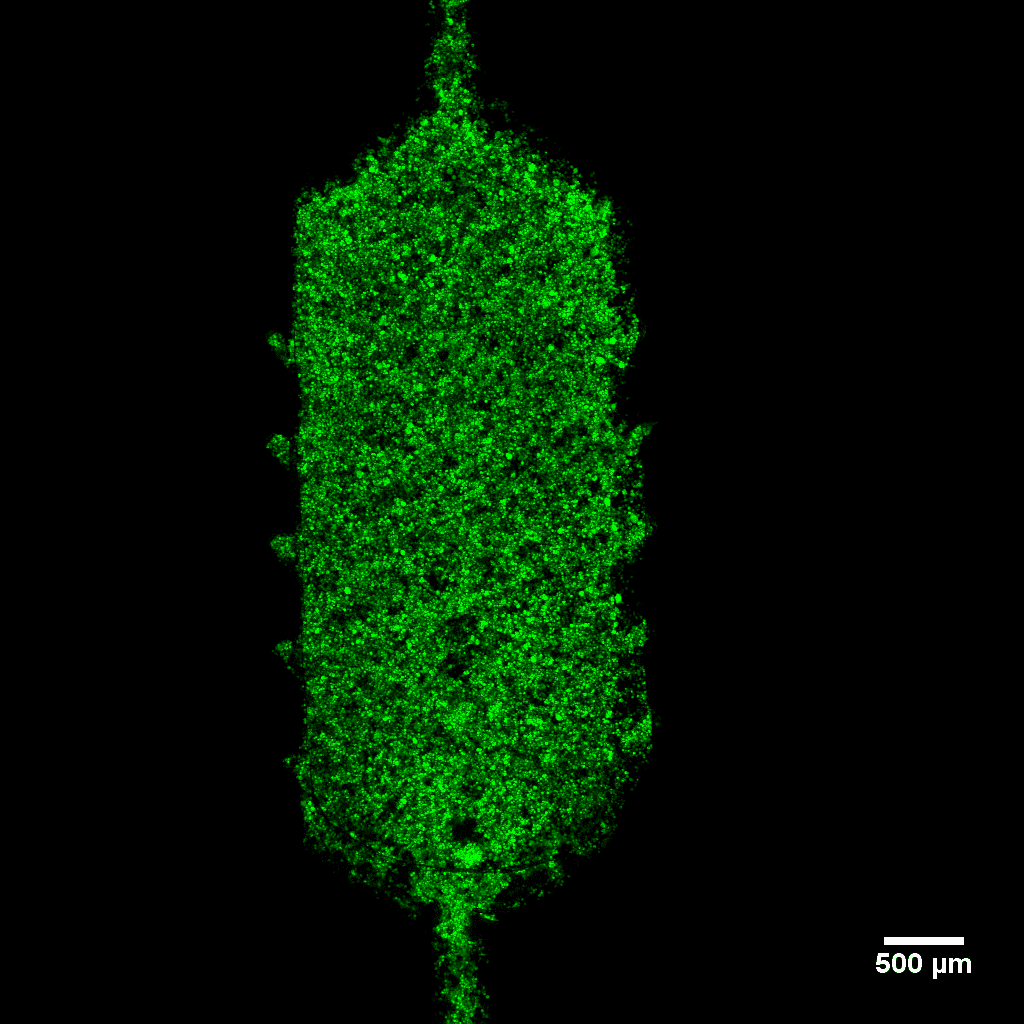
Cyclo olefin polymer (COP) and cyclic olefin copolymer (COC) are our choice material for all our products. These medical-grade plastics are an optimal material for Organ-on-chip and life science applications.

Impermeability
These materials have a very low permeability to oxygen and water vapor, allowing precise control of those gases inside the microchannel by controlling their concentration in the culture medium and its flux. In fact, it is possible to perform hypoxia experiments inside our chips, by carefully controlling those parameters.
No unespecific adsorption issues
Other widely used compounds for OoC production such as PDMS suffer from unspecific absorption making them unusable for drug testing experiments. COP and COC are lipophobic materials that doesn’t present this issue, allowing their use in drug development and diffusion experiments.
Optical properties
COP and COC are outstanding optical properties. These materials offer transparency in the visible and near UV range, low birefringence, high Abbe number, making these materials ideal for microscopy applications.
Good chemical and heat resistance
COP and COC present remarkable chemical resistance to acids and even polar solvents. In addition, these compounds present a high glass transition temperature, in some formulations close to 190 °C.
Mass production
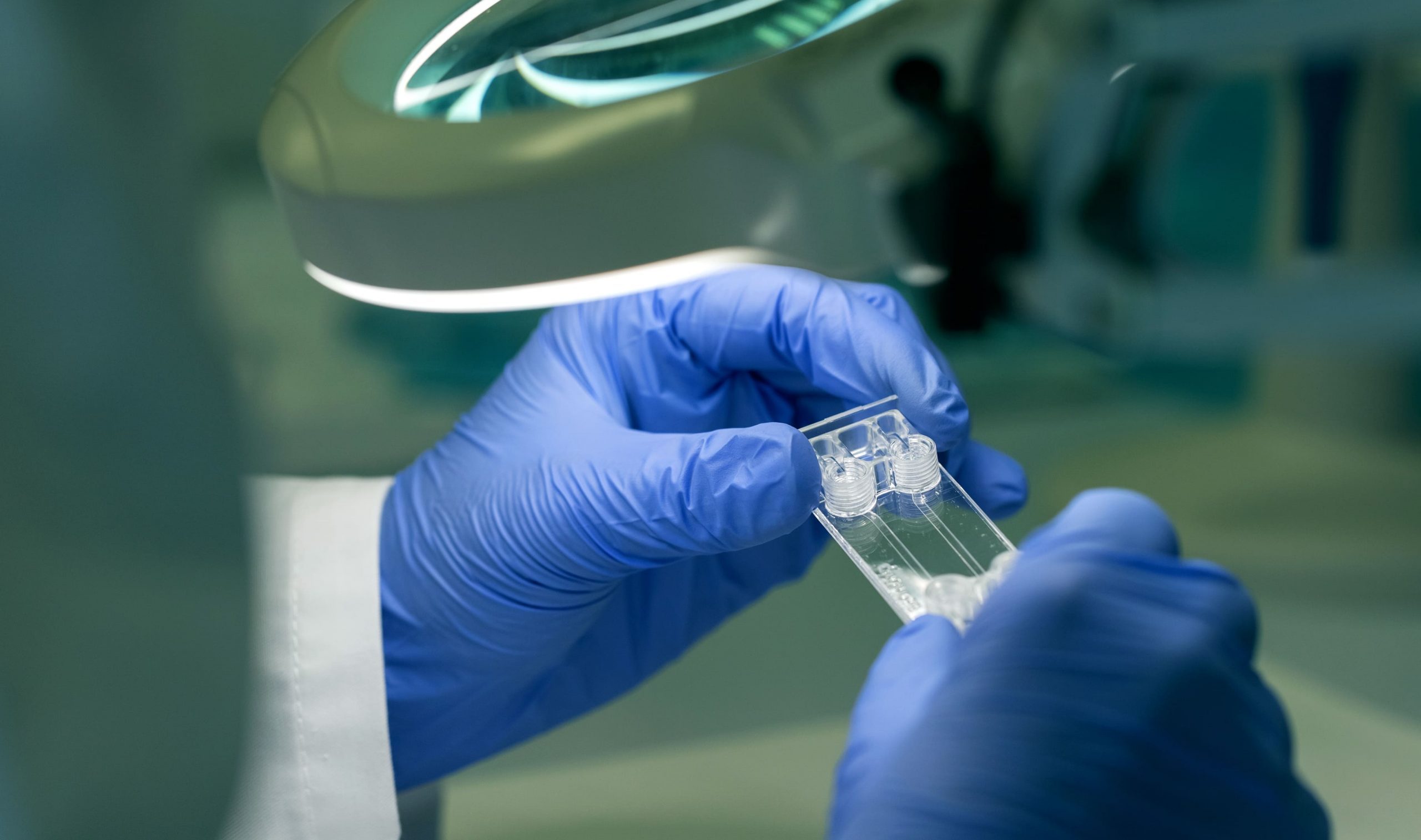
Beonchip’s products are manufactured by thermoplastic injection, assuring reproducibility between batches and allowing high volume production without compromising their quality.

Microfluidic compatibility
Beonchip’s microfluidic connections are designed to be used with any type of microfluidic control system such as syringe pumps, peristaltic pumps or pressure-based flow control systems. We can provide adaptors for our chips, that are compatible with Luer or barb connections of your flow control system.